The last 50 years have seen a steady increase in life expectancy across the developed world mainly due to disease prevention, improved hygiene, and nutrition. However with increasing age, a new problem has appeared - the phenomenon of inflammaging and immune senescence. This is now a focus for research as the decline in immune parameters which can lead to disabilities and poor health in an aging population, has significant impact on social welfare.
Inflammaging is a hallmark of virtually every major age-related disease and phenotype such as dementia, Parkinson’s disease, atherosclerosis, type 2 diabetes, cancer, and arthritis, and is a pathological characteristic of aging tissues found across multiple species (Xu and Larbi 2017).
Contents
Innate Immunity
Granulocytes
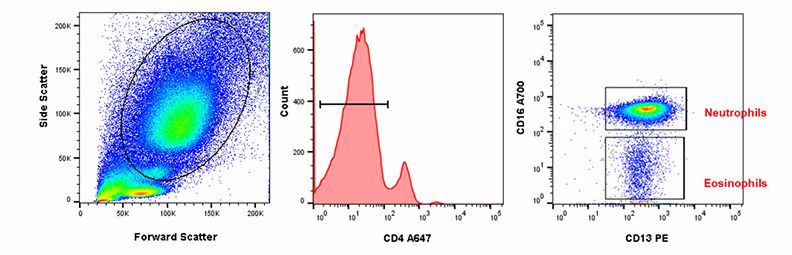
With age, the absolute number of neutrophils does not change, but they do have reduced capability to migrate to chemotactic signals such as IL-8 and fMLP. A reduced ability of chemotaxis could also lead to reduced egress of neutrophils from inflamed tissue leading to higher local inflammation. It has been reported that neutrophils have lower levels of surface CD16, which can lead to lower superoxide generation and reduced capacity to uptake opsonized particles. Recently the ability to form neutrophil NETs has been shown to decrease with aging which may be one of the reasons for increased susceptibility to infections in the elderly (Pinti et al. 2016). Figure 1 below shows how staining with CD4, CD16, and CD13 allows identification of neutrophils and eosinophils in human peripheral blood. Visit our dedicated granulocyte page for more information on markers to identify granulocyte subsets and their function.
Fig. 1. Neutrophil and eosinophil staining in human peripheral blood. After using forward and side scatter to identify the myeloid population, CD4 (MCA1267P647) was used to distinguish between granulocytes and monocytes. CD16 (MCA2537A700) and CD13 (MCA1270PE) allow identification of CD16+CD13+ neutrophils and CD16-CD13+ eosinophils within the CD4- population.