Pyroptosis is a form of lytic regulated cell death (RCD) first described by Zychlinsky and colleagues in 1992 as apoptosis, but later renamed as pyroptosis in 2001 by Cookson and Brennan to reflect its inflammatory nature. It is triggered by perturbations of extracellular and intracellular homeostasis in the innate immune system.
Its pro-inflammatory nature distinguishes it from other forms of RCD, like apoptosis and necroptosis, and it is in fact the most immunogenic of all the cell death mechanisms. It has distinct morphological features such as cellular swelling, chromatin condensation, and plasma membrane permeabilization. This mini-review highlights the role pyroptosis plays in regulated cell death and the mechanisms involved.
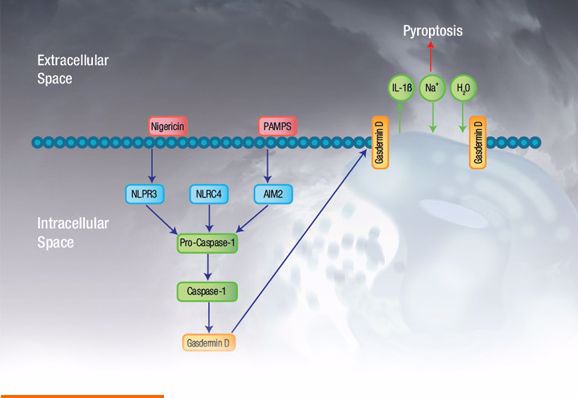
Pyroptosis is mediated by the pyroptotic caspases, caspase-1, caspase-4, and caspase-5 in humans, and caspase-11 in mice. Caspase-1 is activated (not necessarily by autoproteolytic processing), upon activation of various inflammasomes, whereas caspase-4, caspase-5, and caspase-11 are activated upon direct binding of LPS from gram negative bacteria. These active caspases then cleave gasdermin D (although more recently other members of the gasdermin family have also been implicated in pyroptosis), into a 199 amino acid carboxy-terminal fragment and a 242 amino acid amino-terminal fragment (in humans). The amino-terminal of cleaved gasdermin D integrates into the cell membrane to form a pore which directly leads to cell death. A simplified overview of the pyroptosis pathway is shown in Figure 1.
Fig. 1. Pyroptosis overview. Simplified pyroptosis pathway showing how signal initiation leads to inflammasome activation, caspase-1 activation, and ultimately gasdermin D associated pore formation and cell death.